 A metallic bond is a chemical bond, in which the atoms do not share or exchange electrons to bond together. The electrons are then part of both atoms and both shells are filled. Valence electrons are shared when an atom needs electrons to complete its outer shell and can share those electrons with its neighbor. A covalent bond is a chemical bond formed by shared electrons. 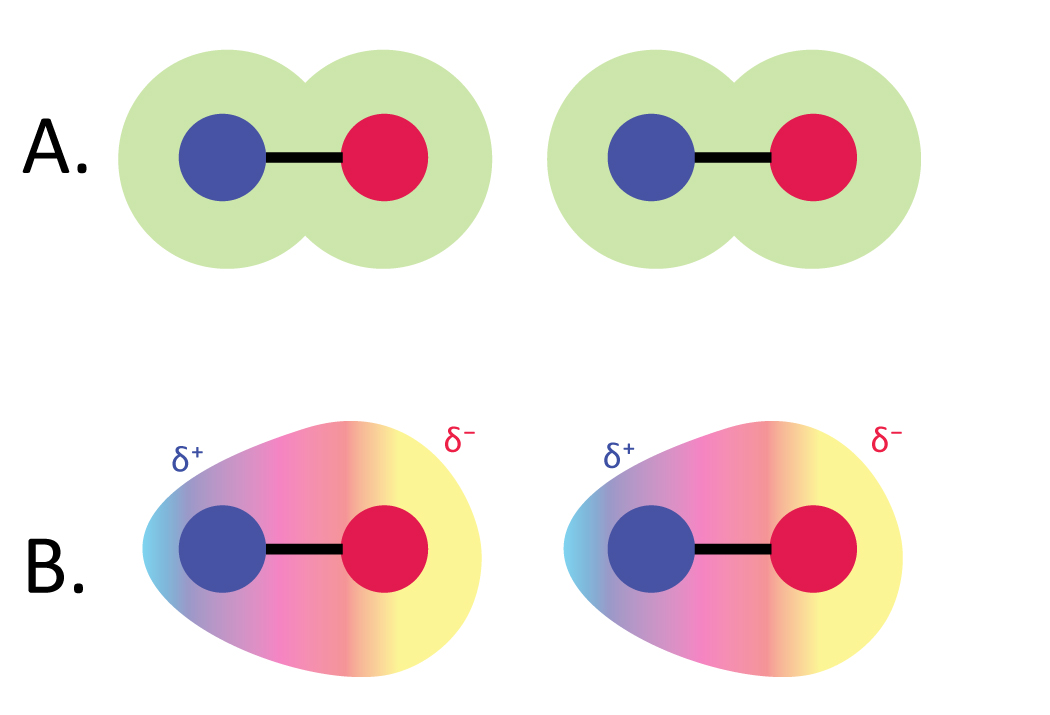
This type of chemical bond is typical between elements with a large electronegativity difference. An ionic bond is a chemical bond, in which one or more electrons are wholly transferred from an atom of one element to the atom of the other, and the elements are held together by the force of attraction due to the opposite polarity of the charge. Therefore, the electromagnetic force plays a major role in determining the internal properties of most objects encountered in daily life. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds. 
The strength of chemical bonds varies considerably there are “primary bonds” or “strong bonds” such as ionic, covalent and metallic bonds, and “weak bonds” or “secondary bonds” such as dipole–dipole interactions, the London dispersion force and hydrogen bonding.Ī chemical bond is a lasting attraction between these atoms, ions or molecules that enables the formation of chemical compounds. Three different types of primary or chemical bond are found in solids. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
